As per ICH Q9 “Quality Risk Management is a systematic process for the assessment, control, communication and review of risks to the quality of the product throughout its life cycle.” There are many methods and tools to perform Quality Risk…


May 1, 2020 Central Drugs Standard Control Organization (CDSCO), the Government of India Drug Regulatory Agency has extended the validity of WHO GMP/ Certificate of Pharmaceutical Product (COPP) expiring from March 2020 to August 2020 by six months from…
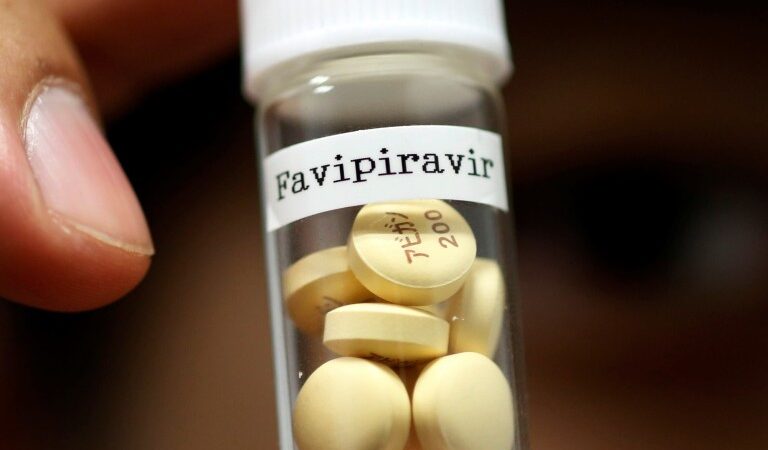
April 30, 2020 Glenmark Pharmaceuticals announced that it has received the Drugs Controller General (I) permission to conduct clinical trial on Favipiravir. As on date, Glenmark is the first pharmaceutical company in India to be given an approval by…

April 28, 2020 The International Coalition of Medicines Regulatory Authorities (ICMRA) on Tuesday has stated “We have stepped up our global collaboration to facilitate and expedite the development and evaluation of diagnostics and therapeutics, including possible vaccines, against SARS-CoV2”. ICMRA…

Any generic industry thrives on defects reduction, increasing productivity, quality improvement, cost reduction, continual improvement etc. There are various principles or methods available, which helps to achieve our aim of Lean Manufacturing Procedures and Good Manufacturing Practices. We all know…
Recent Post
- CDSCO Issues Public Notice on Disposal of Long-Pending SUGAM Applications
- India Bans High-Dose Nimesulide: What You Need to Know
- Compounding of Offences under the Drugs and Cosmetics Act, 1940: What Stakeholders Need to Know
- 🚨 CDSCO Directive on Immediate Inspections as per Revised Schedule M – Is Your Facility Ready?
- CDSCO Issues Regulatory Clarification on Combi-Pack Approvals: Key Insights for Injectable Manufacturers

.png
)