5th March, 2025 The Ministry of Health and Family Welfare, Government of India, has taken a significant step to streamline and enhance the registration process for Clinical Research Organizations (CRO) by mandating online registration through the SUGAM portal. This initiative,…


Ethics Committee is a body responsible for ensuring that medical experimentation and human subject research are carried out in an ethical manner in accordance with national and international laws. Central Drugs Standard Control Organization, the national drug regulatory authority has…
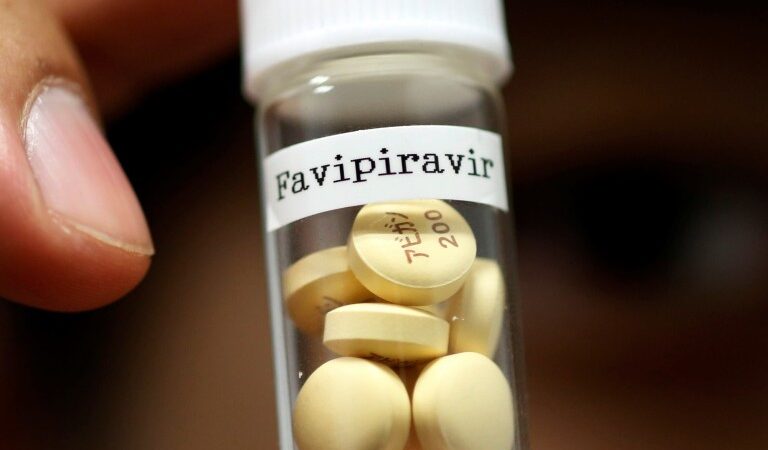
April 30, 2020 Glenmark Pharmaceuticals announced that it has received the Drugs Controller General (I) permission to conduct clinical trial on Favipiravir. As on date, Glenmark is the first pharmaceutical company in India to be given an approval by…

April 18, 2020 Central Drugs Standard Control Organization (CDSCO), the Government of India Drug Regulatory Agency has permitted the conduct of clinical trial of Convalescent Plasma In COVID-19 Patients. The trial protocol was developed and submitted by Indian Council of…
Recent Post
- CDSCO Issues Public Notice on Disposal of Long-Pending SUGAM Applications
- India Bans High-Dose Nimesulide: What You Need to Know
- Compounding of Offences under the Drugs and Cosmetics Act, 1940: What Stakeholders Need to Know
- 🚨 CDSCO Directive on Immediate Inspections as per Revised Schedule M – Is Your Facility Ready?
- CDSCO Issues Regulatory Clarification on Combi-Pack Approvals: Key Insights for Injectable Manufacturers

.png
)