Solubility is the capacity of the solvent to dissolve a solute. Solutes may differ in both the extent and the rate at which they dissolve in a solvent. The solubility of one substance in another is a measure of the…


- admin
- 2 Comments.
- GMP Guidelines
Glass containers used in pharmaceutical preparations are intended to come into direct contact with pharmaceutical preparations. Glass containers shall have a proper closure system. The containers are designed in a manner that the removal of contents is hassle free and…
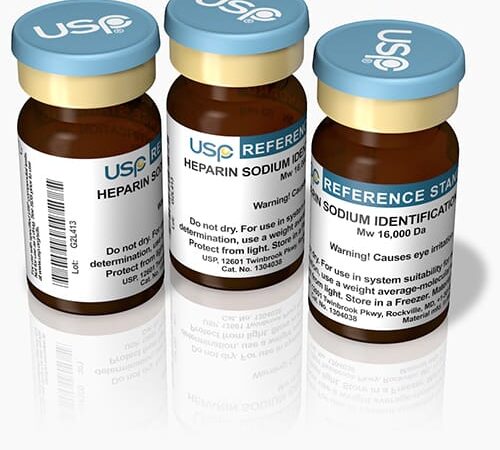
Testing of identity, strength, quality and purity of pharmaceuticals such as drug substances, excipients and products requires them to be compared with a substance which is highly purified and characterized. The US Pharmacopeia (USP) defines reference standard materials as "highly…

Parametric release is defined as the system of release that gives the assurance that the product is of the intended quality based on information collected during the manufacturing process and on the compliance with specific GMP requirements related to Parametric…
Recent Post
- CDSCO Issues Public Notice on Disposal of Long-Pending SUGAM Applications
- India Bans High-Dose Nimesulide: What You Need to Know
- Compounding of Offences under the Drugs and Cosmetics Act, 1940: What Stakeholders Need to Know
- 🚨 CDSCO Directive on Immediate Inspections as per Revised Schedule M – Is Your Facility Ready?
- CDSCO Issues Regulatory Clarification on Combi-Pack Approvals: Key Insights for Injectable Manufacturers

.png
)